PATIENT EXPERIENCE
Engagement
That Drives Trial ROI
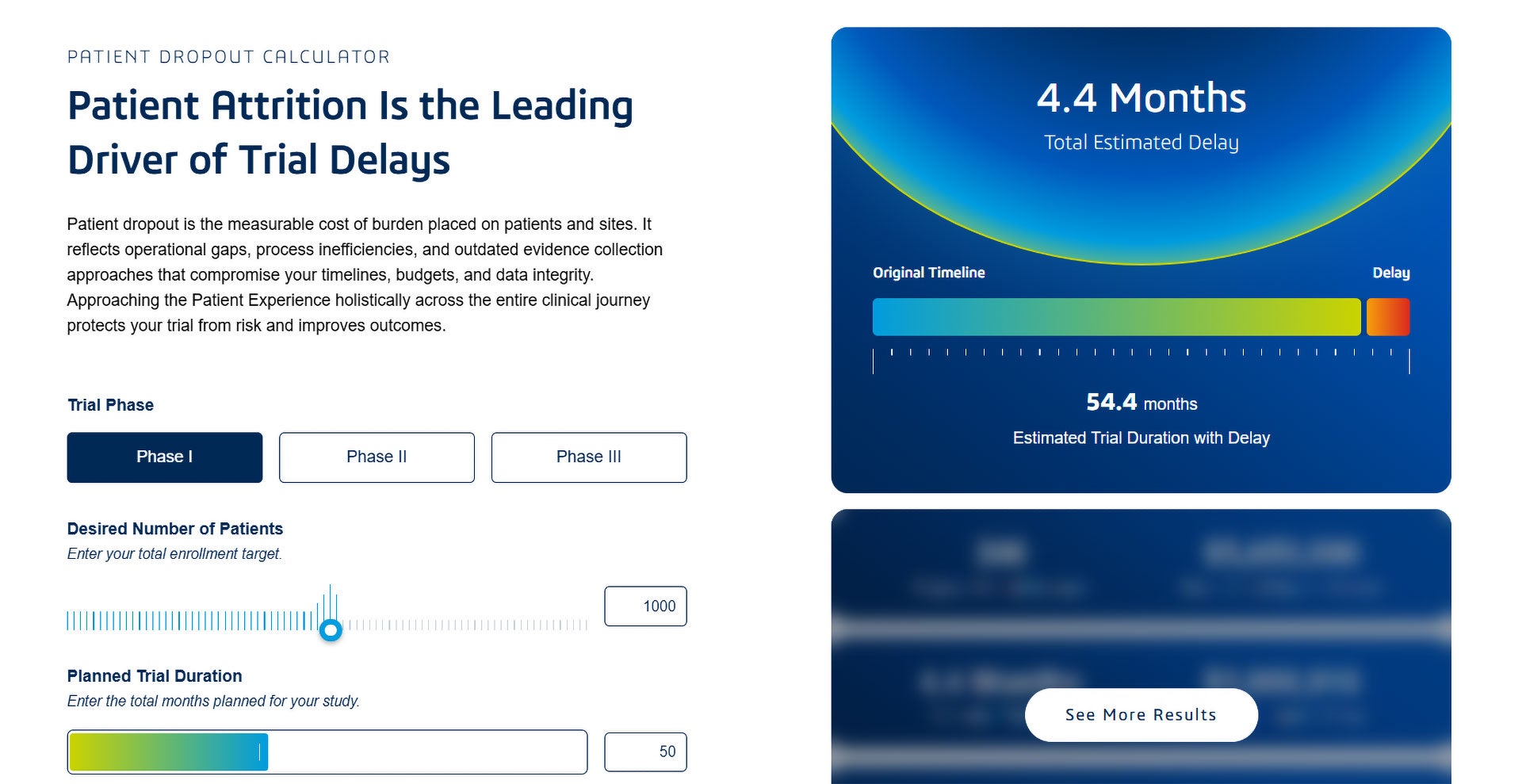
Trials fail because patient participation is harder than it should be.
Repetitive data entry. Disconnected devices. Delayed site follow-up. Each friction point compounds, increasing the risk of dropout.
When you embed the Medidata Patient Experience across the lifecycle, participation becomes predictable, manageable, and visible—from protocol design to post-trial follow-up.
Sign up for Medidata Plus for full access to the Al-layer of the Medidata Platform.
Design & Planning
When feasibility, diversity, and site realities are integrated early, studies advance faster—improving enrollment, strengthening equity, and reducing the need for protocol amendments.
Build & Start‑up
Clear onboarding, streamlined documentation, and connected tools build trust early—driving faster activation, confident enrollment, and stronger retention from the start.
Study Execution
Consistent, simple experiences sustain participation. AI-driven monitoring delivers real-time visibility and early risk detection. Transform your patient engagement from reactive follow-up to predictive oversight.
Decentralized Clinical Trials
Hybrid flexibility without operational fracture
Medidata eCOA
Consistent symptom and outcome reporting
Medidata Sensors
Integrated device data within the same data fabric
myMedidata
Centralized patient portal for visibility and control
Patient Payments
Transparent compensation processes
Registries
Longitudinal engagement beyond episodic visits
Closeout & Post‑trial
The final experience shapes how your patients remember their trial participation—and whether they engage again. Preserve data integrity through submission, close studies clearly, and maintain patient trust.
FAQ
Explore Experiences
Discover the Medidata Platform