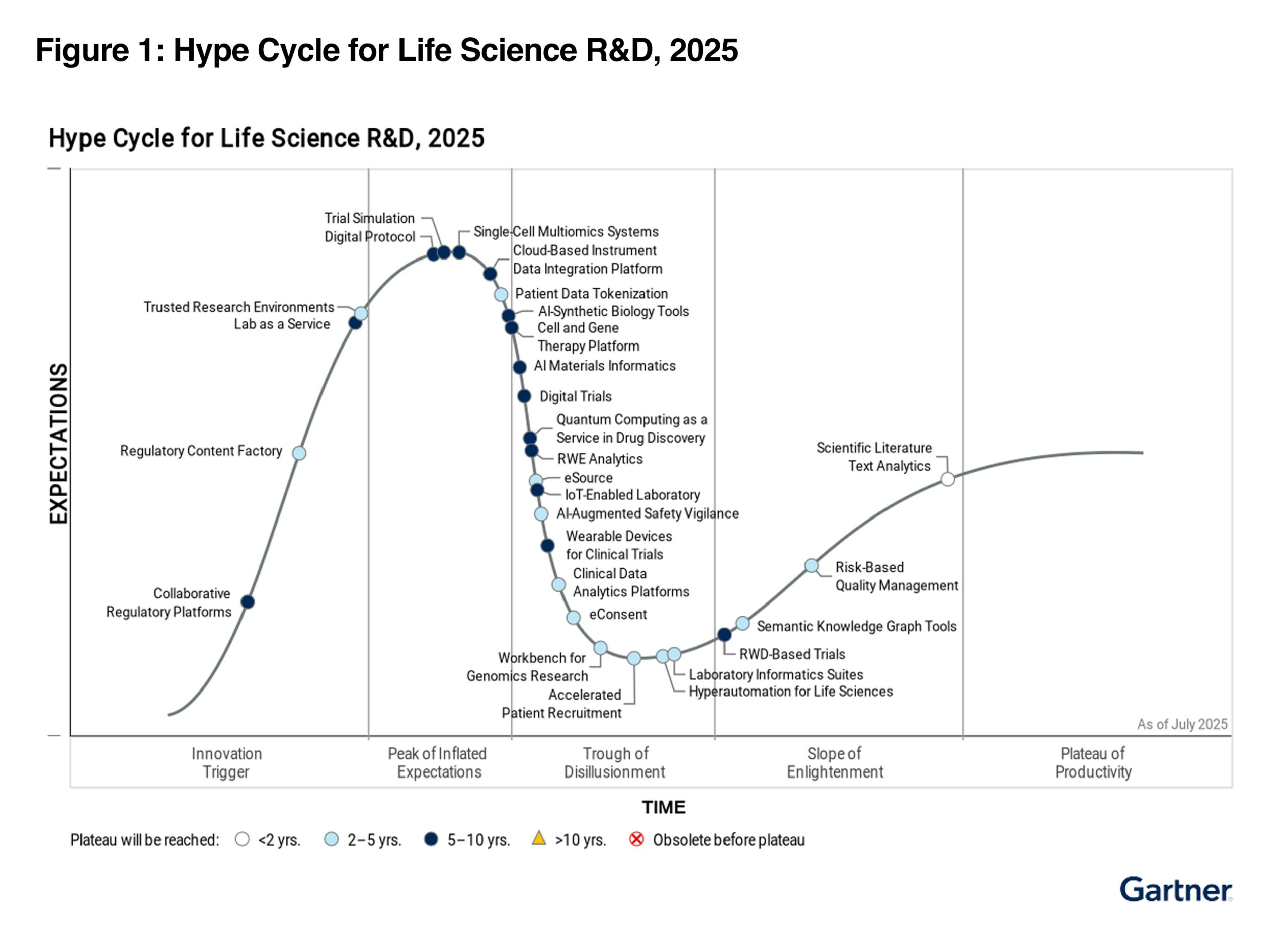
Gartner® Hype Cycle™ for Life Sciences R&D, 2025
Life Science R&D leaders and CIOs are at a critical inflection point, facing immense pressure to differentiate their organizations through strategic adoption of AI, multiomics, and cloud platforms, all while navigating a complex landscape characterized by exponential data growth, regulatory scrutiny, and tight budget constraints to identify and invest in emerging technologies that will deliver true transformational ROI in the shift toward precision medicine.
We believe this strategic Gartner report serves as your definitive roadmap to time your investments, mitigate risk, and scale the innovations that drive true scientific discovery.
Why “Digital Protocol” is the New R&D Cornerstone
Gartner identifies the Digital Protocol as a rapidly evolving innovation approaching the Peak of Inflated Expectations. For the C-suite, this represents the single most important transition in modern research: the move from document-based clinical trials to a structured, data-first workflow.
Download the report to discover:
- The 2026 technology priority investments for Life Science CIOs.
- How to transition from “Document-Centric” to “Data-Centric” R&D.
- Which technologies are maturing and ready for immediate adoption.
Gartner, Hype Cycle for Life Science R&D, 2025, 7 July 2025, Reuben Harwood, Jeff Smith
Gartner and Hype Cycle are trademarks of Gartner, Inc. and/or its affiliates. Gartner does not endorse any company, vendor, product or service depicted in its publications, and does not advise technology users to select only those vendors with the highest ratings or other designation. Gartner publications consist of the opinions of Gartner’s business and technology insights organization and should not be construed as statements of fact. Gartner disclaims all warranties, expressed or implied, with respect to this publication, including any warranties of merchantability or fitness for a particular purpose.
This graphic was published by Gartner, Inc. as part of a larger research document and should be evaluated in the context of the entire document. The Gartner document is available upon request from Medidata.