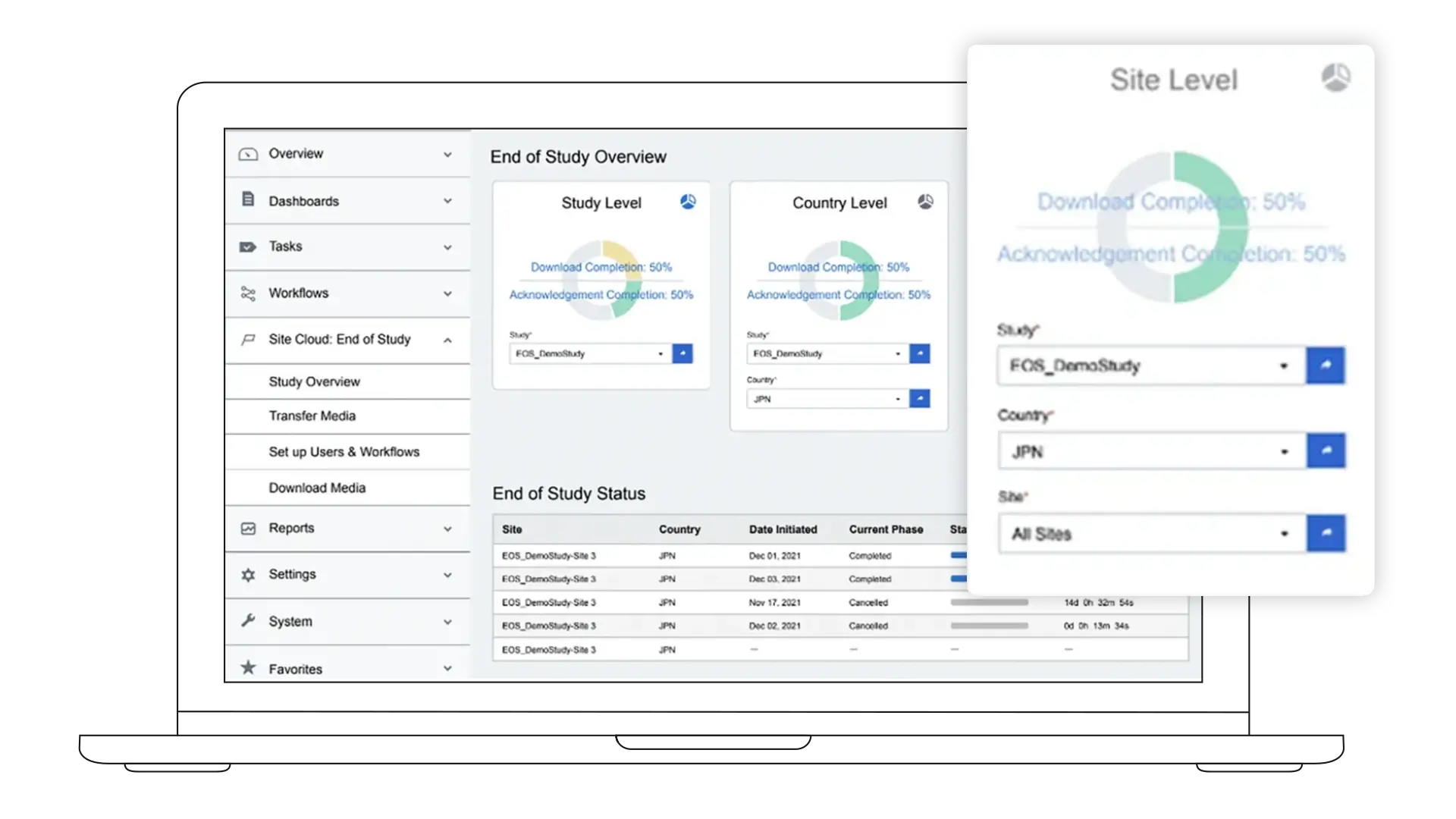
Site Cloud: End of Study
Closing out a study introduces risk when files are scattered, manually shared, or stored on physical media.
Medidata Site Cloud: End of Study (EOS) provides a secure, end-to-end way to generate, distribute, and oversee site study files at trial close. Study files are delivered through a single system that centralizes access, permissions, audit trails, and real-time visibility, bringing control and traceability to study closeout.
Maintain Control
at Study Closeout
Manual handoffs, physical media, and fragmented files create risk at study closeout.
Site Cloud: End of Study replaces that complexity with a controlled, auditable closeout process that keeps end-of-study documentation complete, secure, and inspection‑ready.
Automated Generation and Distribution
Full Audit Trail
Highest Security Standards (SOC 2+)
Improved Site Satisfaction
Electronic Signature for Acknowledgement
One Repository for All EOS Files
Why Choose Site Cloud: End of Study
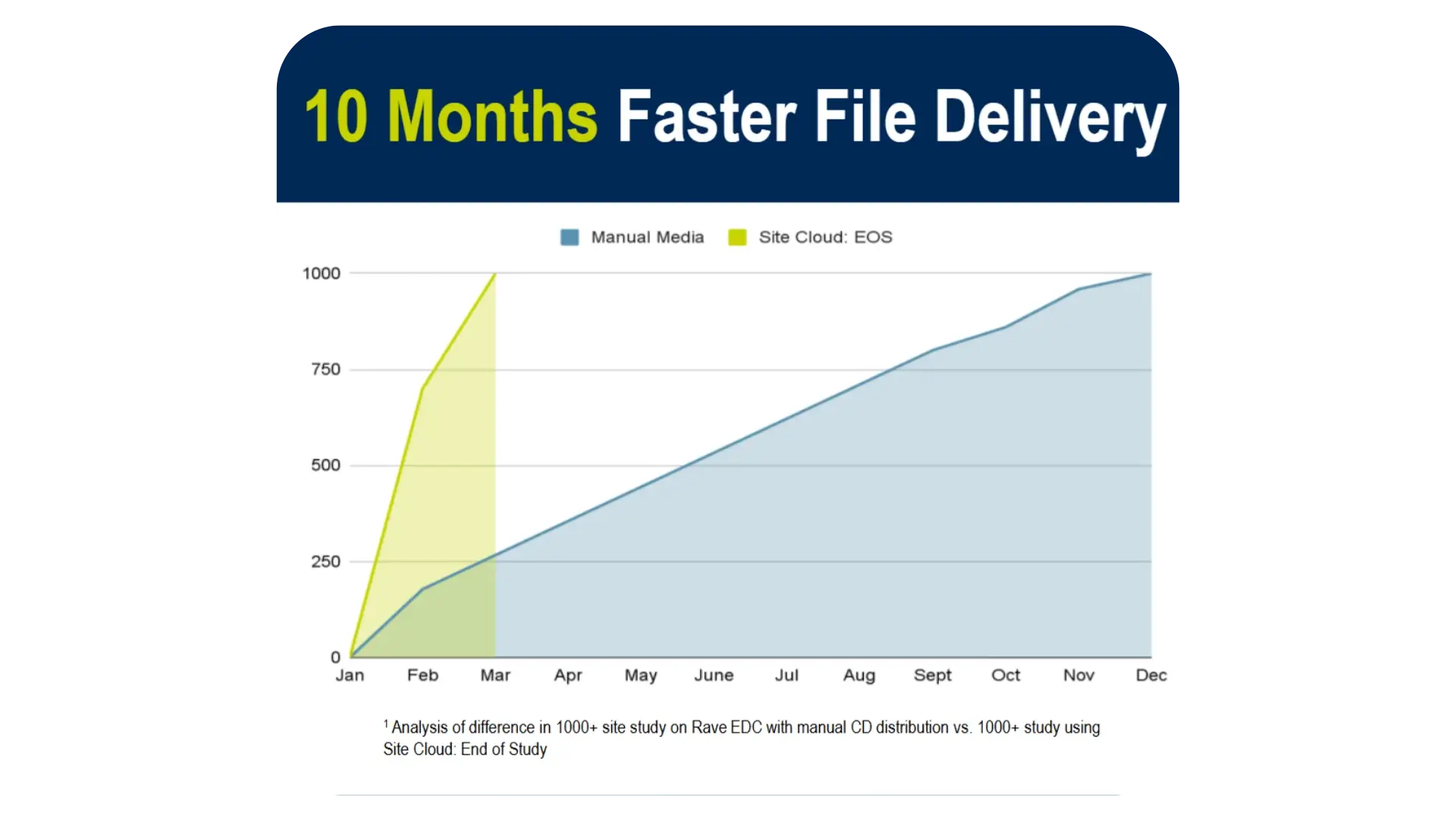
Faster File Delivery
Accelerate Study Closeout
Media is captured in the cloud for streamlined workflows.
Eliminate the time and cost of creating CDs, and gain immediate, secure access to your data.

FAQ
Explore Experiences
Discover the Medidata Platform