myMedidata
Clinical trials ask a lot of patients. Clinical trial participation shouldn’t make it harder.
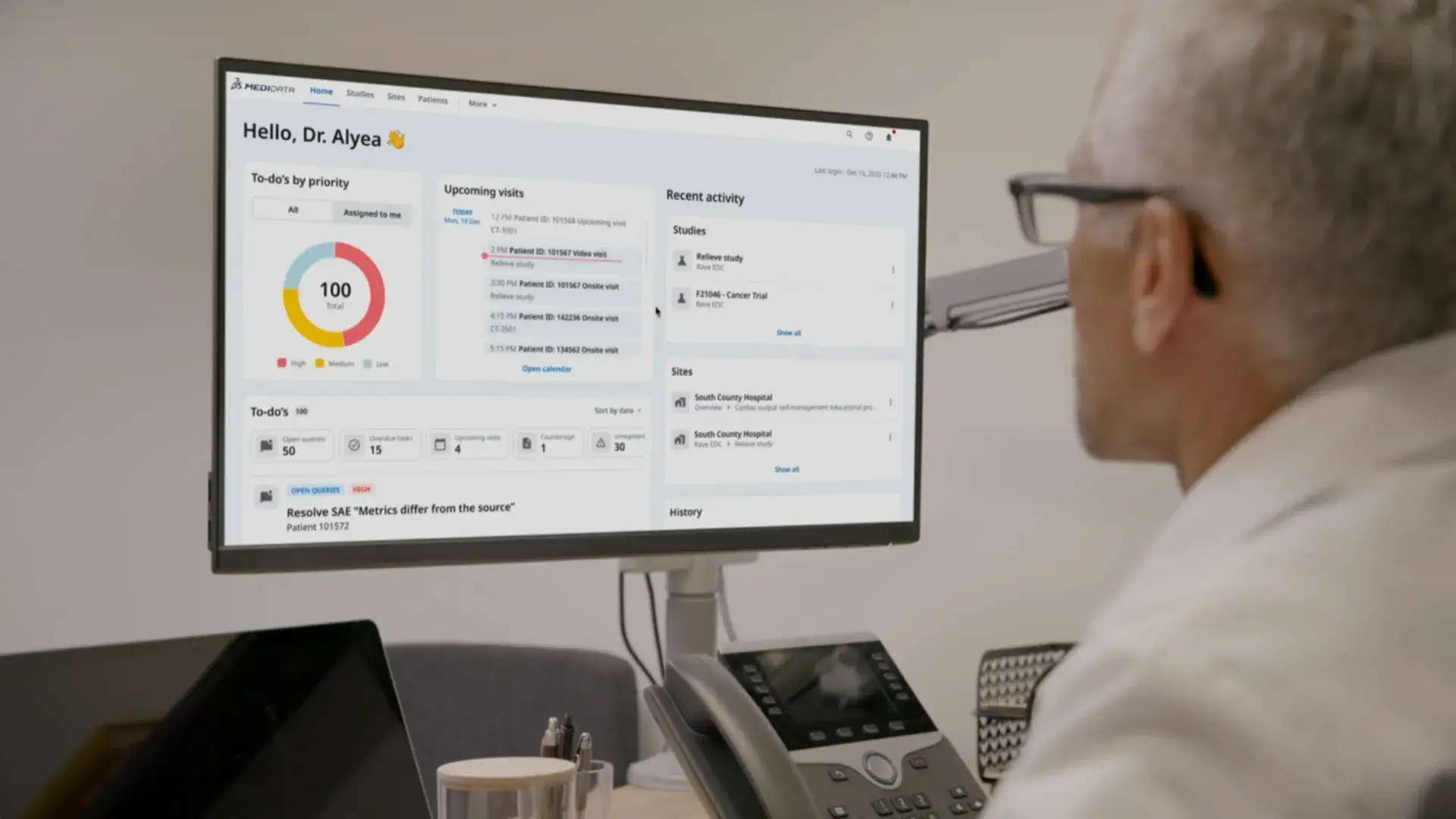
myMedidata is a single patient portal built with patients, for patients. It brings all patient trial activities including eCOA, consent, sensors, and payments into one connected experience, so participation stays consistent as trials evolve.
One Account, Simplified Participation
myMedidata brings patient participation into a single, consistent experience. One account helps reduce friction for patients, simplify coordination for sites, and support retention across the study.
Patient-centric by Design
Device Agnostic
Trials without Boundaries
Keeping Patients and Sites Aligned
Patients
One Place for All Trial Activities
Eliminate extra apps, logins, and unnecessary steps by giving patients one location for all their clinical trial activities.
myMedidata makes it simple for patients to participate and stay engaged throughout their entire trial journey.
What Comes Together in myMedidata
myMedidata brings together Medidata’s patient-facing solutions in a single experience, so participation, communication, and data collection stay connected throughout the study. It creates a consistent experience for patients and a unified source of patient data for study teams.

Featured Resource
The Value of Decentralized Clinical Trials
Hear from Medidata’s CEO on how decentralized and hybrid trial models are changing patient participation, site workflows, and study execution, and what that shift means for sponsors planning future studies.
FAQ
Explore Experiences
Discover the Medidata Platform