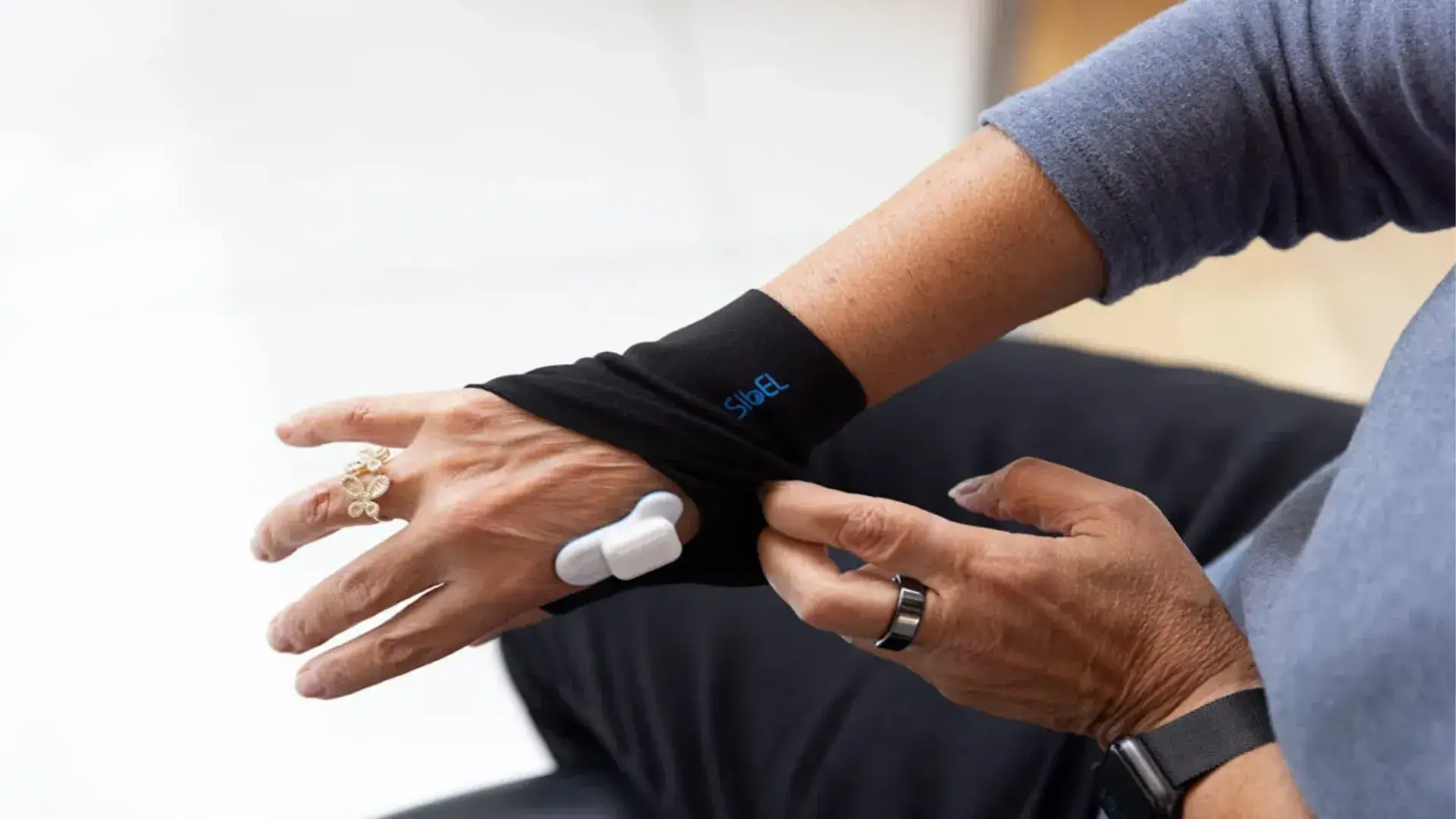
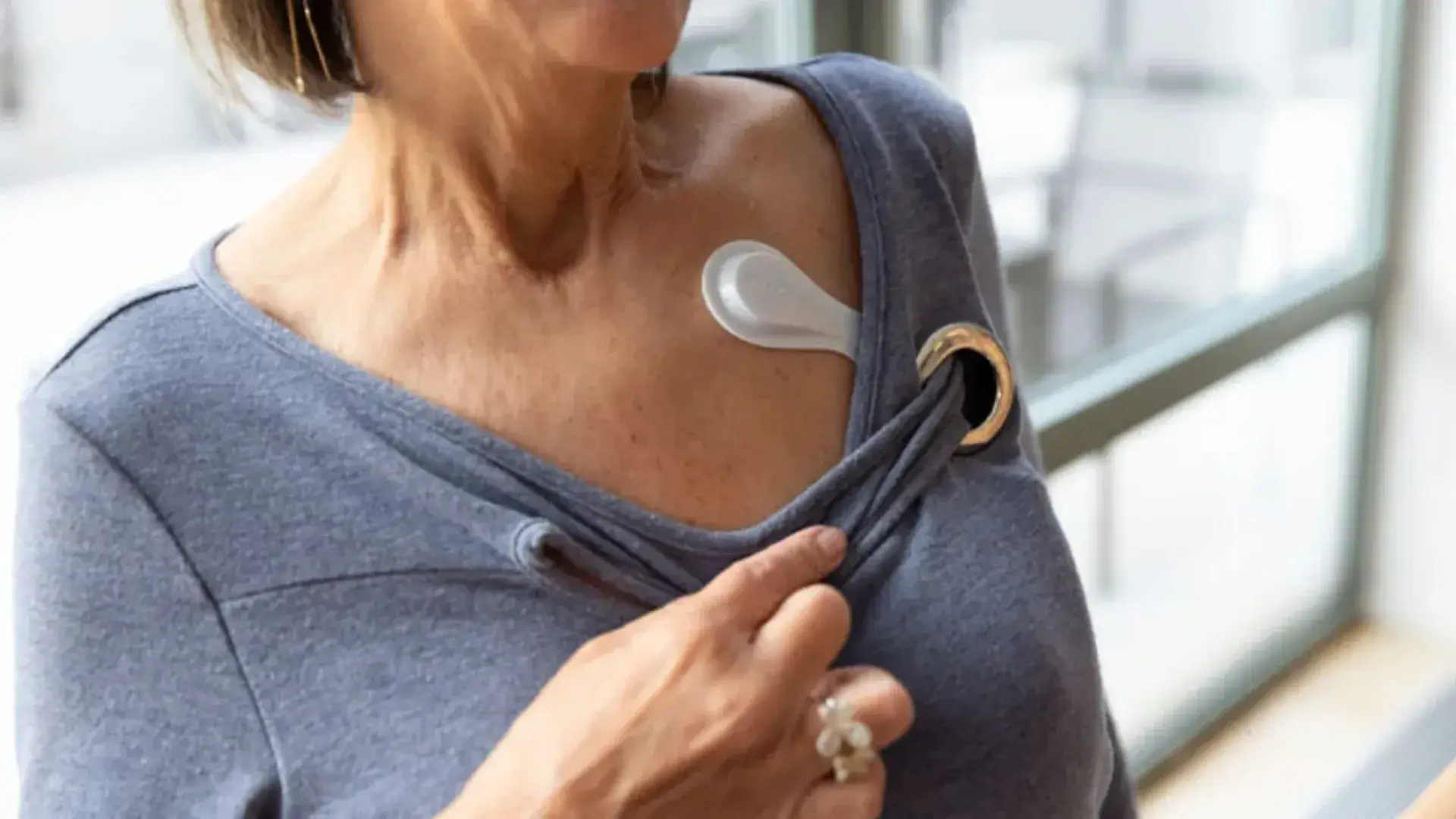
Medidata Sensors
Clinical trials can’t rely on clinic snapshots alone to understand patient health.
Medidata Sensors enables continuous, medical-grade data capture, combining objective digital measures with eCOA to understand how patients feel, function, and respond to treatment over time, without increasing participation burden.
From Continuous Data to Endpoint-ready Evidence
Continuous data only creates value when it forms evidence, not just signals. Medidata Sensors brings objective digital measures and subjective eCOA together within the same study context, using validated algorithms to generate evidence as the study progresses. This approach supports digital endpoints that can be evaluated with greater confidence during the trial.
Enrich Scientific Evidence Generation
Objective and Patient-reported Data Aligned
Earlier Visibility into Emerging Issues
Shared Clinical Context across Study Teams
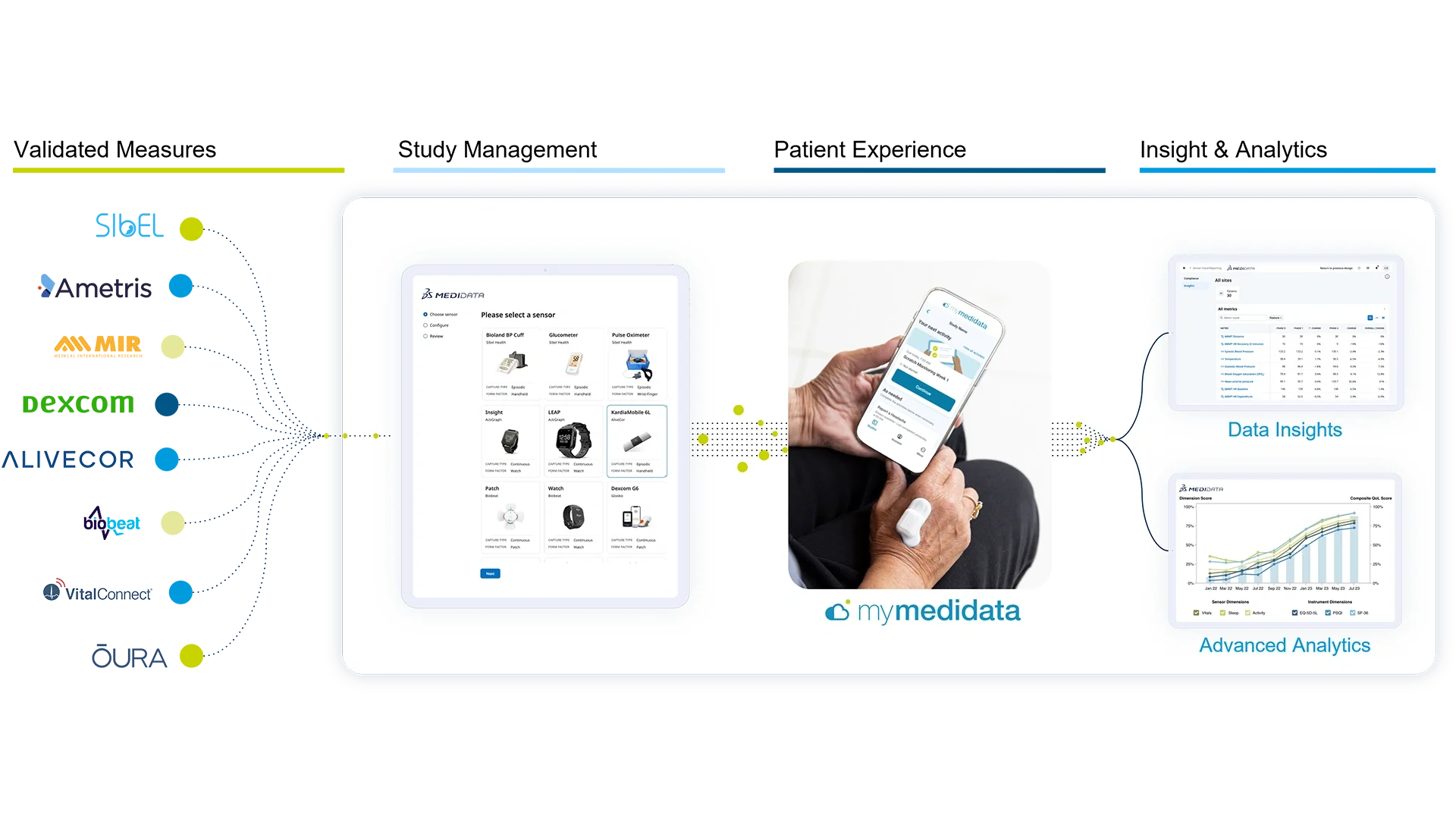
Low-code, No-code Sensor Study Configuration
Trial-grade Sensor Integrations
What Medidata Sensors Supports
Medidata Sensors supports both continuous collection and episodic collection of a broad range of physiological and functional measures used in regulated clinical trials.
Vital Signs
Cardiac
Respiratory
Metabolic
Sleep
Snoring
Scratch
Cough
Electrodermal Activity
Falls
Activity
Mobility
Involuntary Movement
Upper Limb Mobility
Physical Functioning
FDA-cleared sensors
510(k) sensors supported
85+
validated sensor measures
Episodic + Continuous
data capture
Trial-grade
sensor integrations
The Evidence Approach in Practice
Sponsors
Endpoint Confidence
Stop guessing at endpoint decisions. Combine objective digital measures with subjective patient reports, using validated algorithms to generate richer evidence as the study progresses.
Surface meaningful change earlier, reduce late-stage surprises, and evaluate digital endpoints with greater confidence during the trial.
Featured Resource
At-home Six-minute Walk Test
Discover a real-world example of how medical-grade sensors enable at-home functional capacity assessments while generating trial-ready cardiac and motion data.
FAQ
Explore Experiences
Discover the Medidata Platform