AI Is eCOA’s Greatest Leap Forward, But Only If Applied Where It Matters Most
Artificial intelligence (AI) comes up in nearly every conversation now, often with the same idea that it will fundamentally change how studies are run. That may be true. But in a field like clinical trials, change rarely happens all at once. It tends to happen in smaller, more deliberate steps that are usually shaped by a sequence of skepticism, validation, and acceptance. Electronic clinical outcome assessment (eCOA) has followed that pattern for years. And that’s why when it comes to AI in eCOA, the more important question is not what it can do, but where it actually delivers true value today.
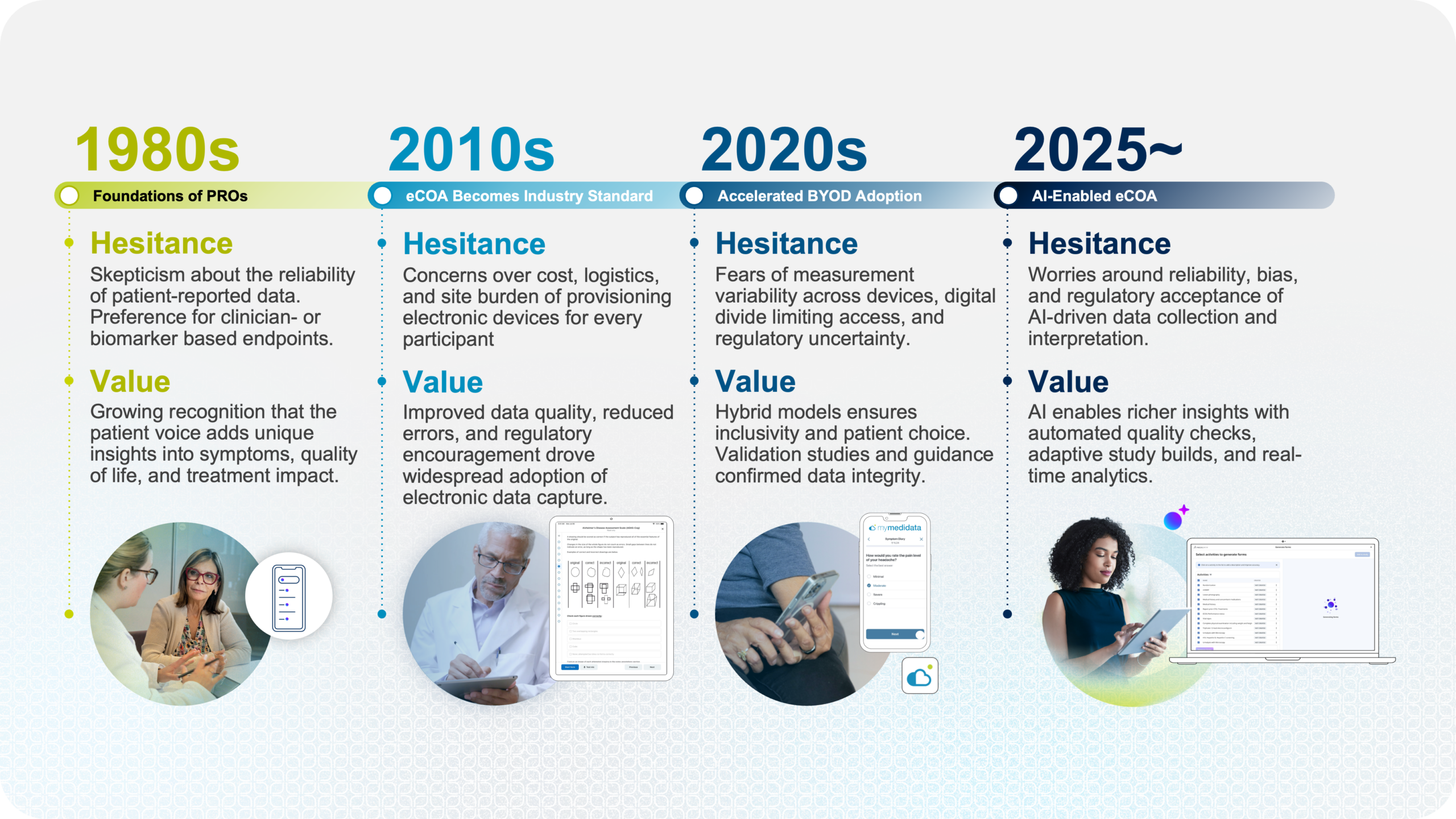
The Life Cycle of eCOA Evolution
There was a time when patient-reported outcomes (PRO) weren’t widely trusted. The idea that subjective inputs from patients could meaningfully inform treatment evaluation wasn’t immediately accepted. Over time, that changed not because the industry was convinced by the possibility, but because it was convinced by evidence. The same thing happened when trials moved from paper to ePRO/eCOA. Concerns around data integrity and equivalence slowed adoption, but eventually gave way to broader acceptance as the benefits became clear.
None of these shifts was driven by excitement alone. They moved forward because they solved real problems in a way that held up under scrutiny—and AI is entering that same cycle.
Navigating the Tension Between Possibility and Reality of AI in eCOA
AI is impressive. With minimal inputs, it can draft copies, create workflows, and structure information that would have taken a huge amount of effort not so long ago. But clinical trials don’t operate in a vacuum. It’s a tightly controlled and regulated process that limits where AI can have practical, real-value applications. For example, in theory, AI could generate new COA questionnaires by simply giving it a prompt. In practice, that approach doesn’t align with how clinical trials operate today. Many instruments are validated, have owners, and require licensing as well as review/approval to deploy in clinical trials. This realization will shift the focus from hype to more grounded, innovative ways to adopt AI in eCOA that delivers true value.
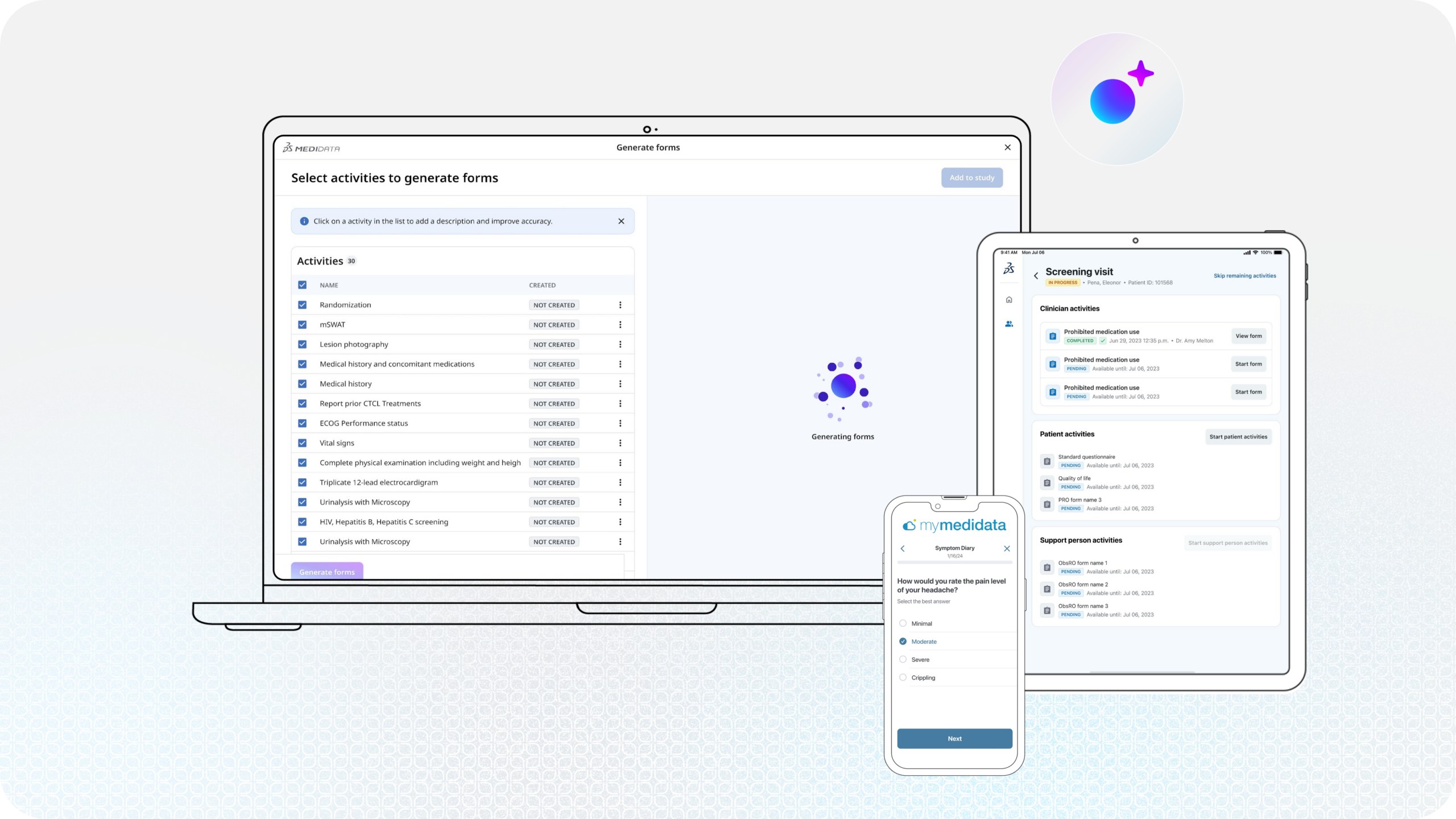
If you take a step back and look at the full lifecycle of eCOA, one part stands out as both complex and overlooked: study build. It’s where protocol is translated into something executable. Every assessment, schedule, and configuration ultimately traces back to how the study was built before it gets implemented for sites and patients. It’s also where a large amount of effort is concentrated and traditionally, much of that effort is manual. Teams review protocols, interpret requirements, and configure systems piece by piece. It’s detailed work that requires precision—and even then, variability can creep in. An already time-consuming process leaves little-to-no room for error to ensure the timeline to first-patient-in is met. This is where AI begins to have meaningful potential for eCOA.
Study Build Is Where AI Starts to Make Sense
The introduction of low-code and no-code tools has already shifted how eCOA studies are built, reducing some of the manual burden associated with custom code and making the process more efficient. AI builds on that progress not by replacing existing workflows, but by targeting the parts of a study build that are inherently repetitive, time-consuming, and prone to variability. Importantly, these tools still operate with a human-in-the-loop, where expertise and oversight remain essential to ensure accuracy and compliance.
One example of an AI-use case is automated UAT test script generation and text execution. One of the biggest challenges with eCOA is it touches many surfaces, including: patients using a web portal, clinicians using a device on site, and data transfer to an electronic data capture (EDC) system—just to name a few of the many scenarios for which the UAT has to cover.
Imagine where AI can automate the generation of test scripts and execute on those scenarios. We’re at a stage where this is possible, having immense potential in expediting timelines and significantly reducing the study build effort. And this is just scratching the surface of what AI can do for eCOA.

Looking Forward With Intentionality
No doubt, AI will continue to expand into other areas of eCOA outside of study build. Patient engagement, smart error detection, and new forms of data analytics all represent potential next steps. But those areas come with additional complexity. They introduce new questions around how data is handled, how engagements are shaped, and how outcomes are interpreted. If the past is any indication, those questions will take time to answer. That doesn’t slow progress though, it simply ensures that when progress happens, it holds.
It’s tempting to think about AI as something that will transform everything at once. But in practice, its impact is likely to be more focused. It’ll show up first in places where it fits naturally; and in eCOA, study build is one of those places.

We’ve only scratched the surface of Medidata’s vision and perspectives for AI in eCOA clinical trials. Discover how AI can fit into your eCOA strategy:
Contact Us

