Medidata TSDV
More than 50% of site monitoring budgets are spent on source data verification (SDV). Yet verifying every data field rarely changes outcomes.
Medidata TSDV (Targeted Source Data Verification) focuses verification on critical safety and efficacy data in Rave EDC, helping you reduce unnecessary effort without compromising regulatory confidence.
SDV Built around Risk
TSDV turns SDV into a structured, risk-aligned strategy. You define what requires verification, at what level, and where, aligned to study-level risk assessments.
Built inside Rave EDC, it enables study- and site-specific SDV strategies while maintaining full coverage of critical safety and efficacy data.
Critical Data Focus
Inspection
Readiness
Study- and Site- Specific SDV
Field-level Verification
Reduced SDV Strategy in Practice
Lower Monitoring Cost
Control SDV-driven Monitoring Costs
Move away from 100% SDV and apply verification only where risk strategy requires it. By reducing unnecessary verification activity and site visits, you lower monitoring costs without compromising oversight.
Download Fact Sheet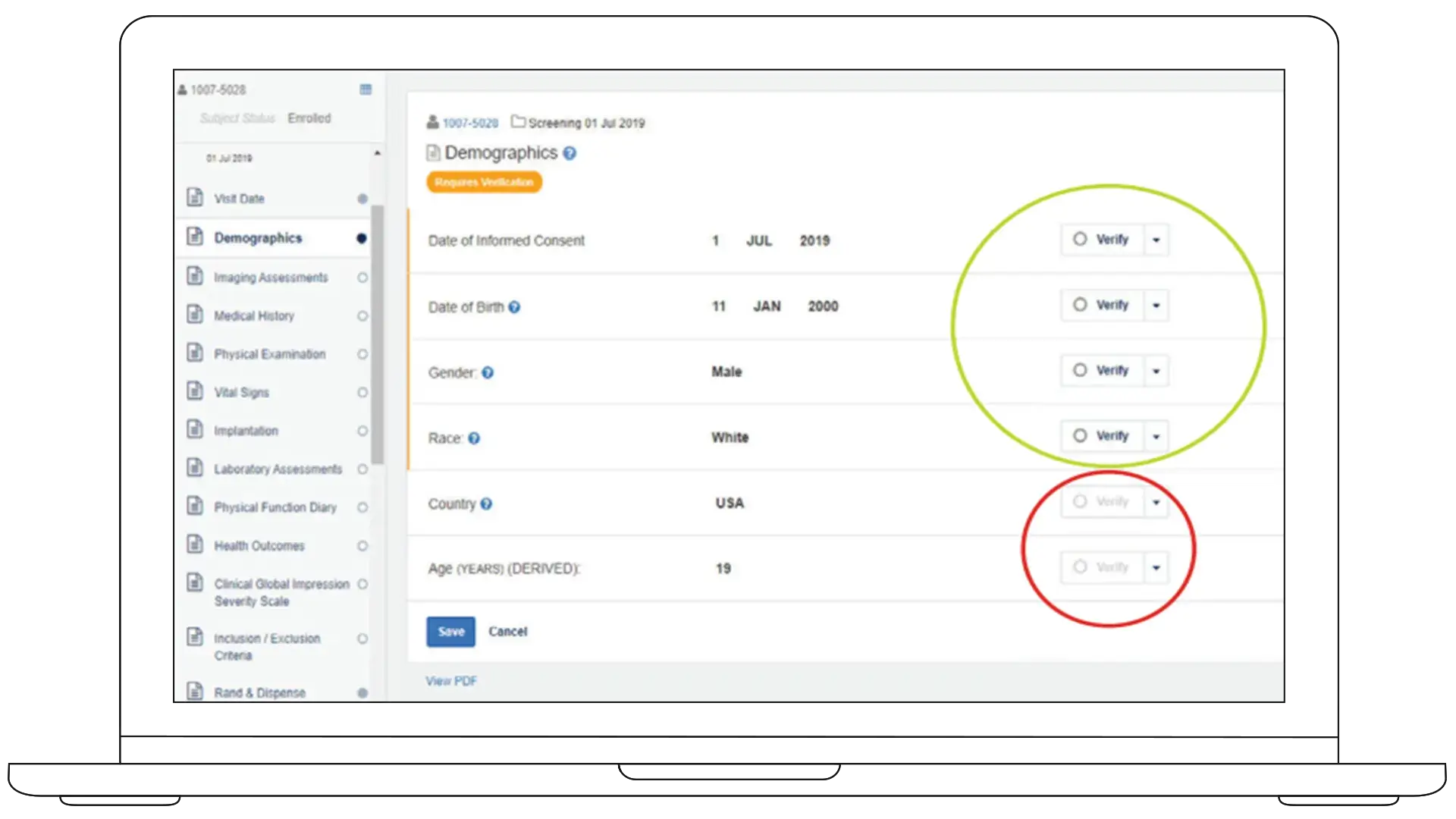
Featured Resource
From 50% to Targeted 15–20% SDV
Nordic Bioscience replaced manual SDV tracking with Medidata TSDV and piloted true risk-based monitoring. The result: streamlined execution and a path toward 15–20% SDV with significant cost savings potential.
Read Case Study
FAQ
Explore Experiences
Discover the Medidata Platform


