Designer
Study build tools have improved, but trial timelines are still under pressure. The real delays don’t happen during initial setup. They show up later, when protocol changes trigger reviews, rework, and uncertainty across the study.
Medidata Designer is a study build solution designed to keep protocol intent, study configuration, and downstream execution connected as change happens.
Design Studies to Keep Up with Change
Built to go beyond initial setup, Designer connects protocol intent to study configuration so updates are defined once and managed consistently as studies evolve.
By centralizing how changes are handled, teams reduce rework, maintain alignment, and keep studies moving from start-up through closeout.
Protocol-aligned Builds
Centralized Study Updates
Reduced Downstream Rework
Clear Version Traceability
Stay in Control as Studies Evolve
Built for Change
Keep Studies Aligned as Protocols Evolve
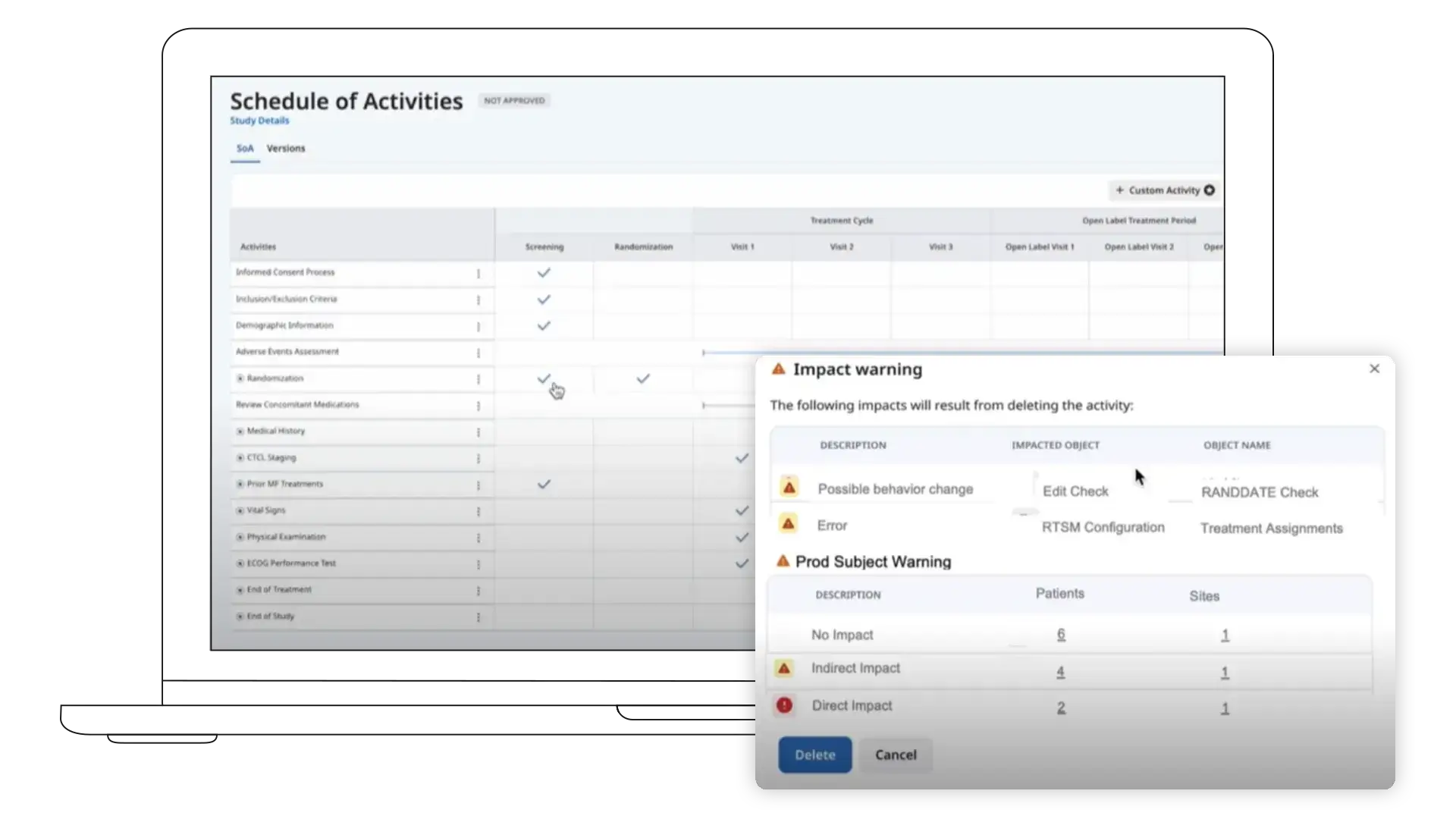
Designer manages study configuration as a single source of truth. When protocols change, updates are applied once instead of rebuilt across forms and systems.
This helps teams manage amendments without fragmenting study structure.
Featured Resource
Move from Protocol to Study Build Faster with AI
See how Medidata Designer removes friction across study build and change, from protocol intake through validation.
View Infographic
FAQ
Explore Experiences
Discover the Medidata Platform