Medidata CTMS
When trial oversight lives in spreadsheets and follow-ups, teams lose critical time and visibility.
Medidata Clinical Trial Management System (CTMS) brings clinical and operational activity into one connected view, working alongside the systems teams rely on to plan, track, and oversee studies.
Where Clinical and Operational Work Come Together
Replace fragmented trackers with a single system designed to reduce clicks, surface what matters most, and keep teams informed through automatic notifications. Enter information once and reuse it seamlessly across workflows.
Visit Monitoring
Issue Management
Oversight and Reporting
Document Submission
and Tracking
ICF Review
Study Management
Why Choose Medidata CTMS
Scalable
Scalable
Reliably support growing portfolios with consistent performance as studies scale.
Download Fact Sheet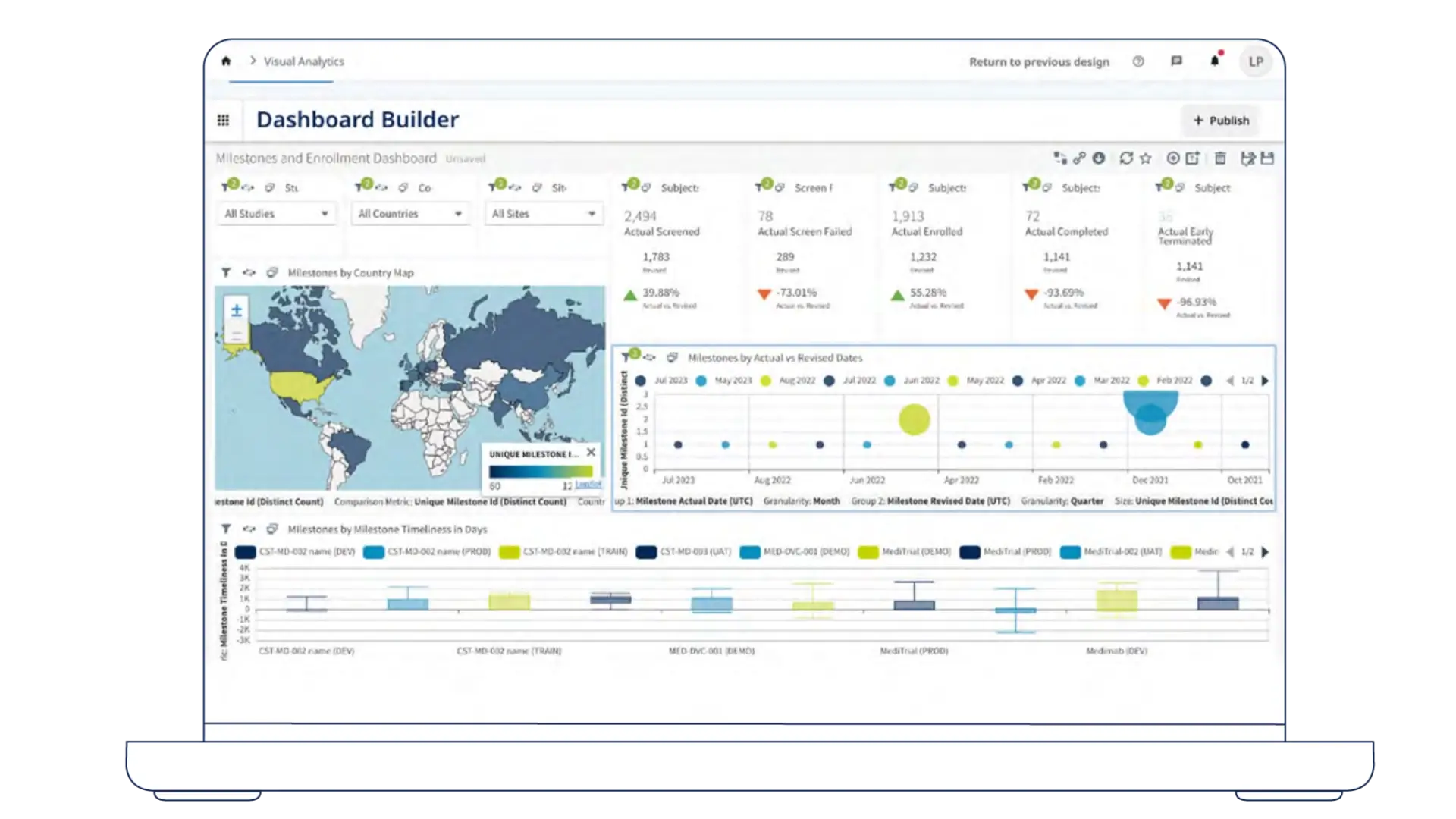
Proven Results with Medidata CTMS
Automated Workflows Make a Lean Team More Efficient
Download Case StudyCASE STUDY
Learn how fast-growing pharmaceutical company Enterin implemented Medidata CTMS to enable their resource-constrained team to work more effectively.
"By automating manual workflow and bringing all of our data together, Medidata CTMS has streamlined our entire clinical trial management lifecycle."

INDUSTRY RECOGNITION
Among thirteen CTMS products, the Everest Group PEAK Matrix® assessment named Medidata CTMS the leading solution in the industry.
Medidata CTMS was recognized as the industry leader in Everest Group's 2024 PEAK Matrix® Life Sciences Clinical Trial Management System (CTMS) Products.


FAQ
Explore Experiences
Discover the Medidata Platform



