Grants Manager
Site budgets break down when planning and negotiation live across disconnected spreadsheets, emails, and systems. Timelines slip, decision-making falters, and sites carry the burden.
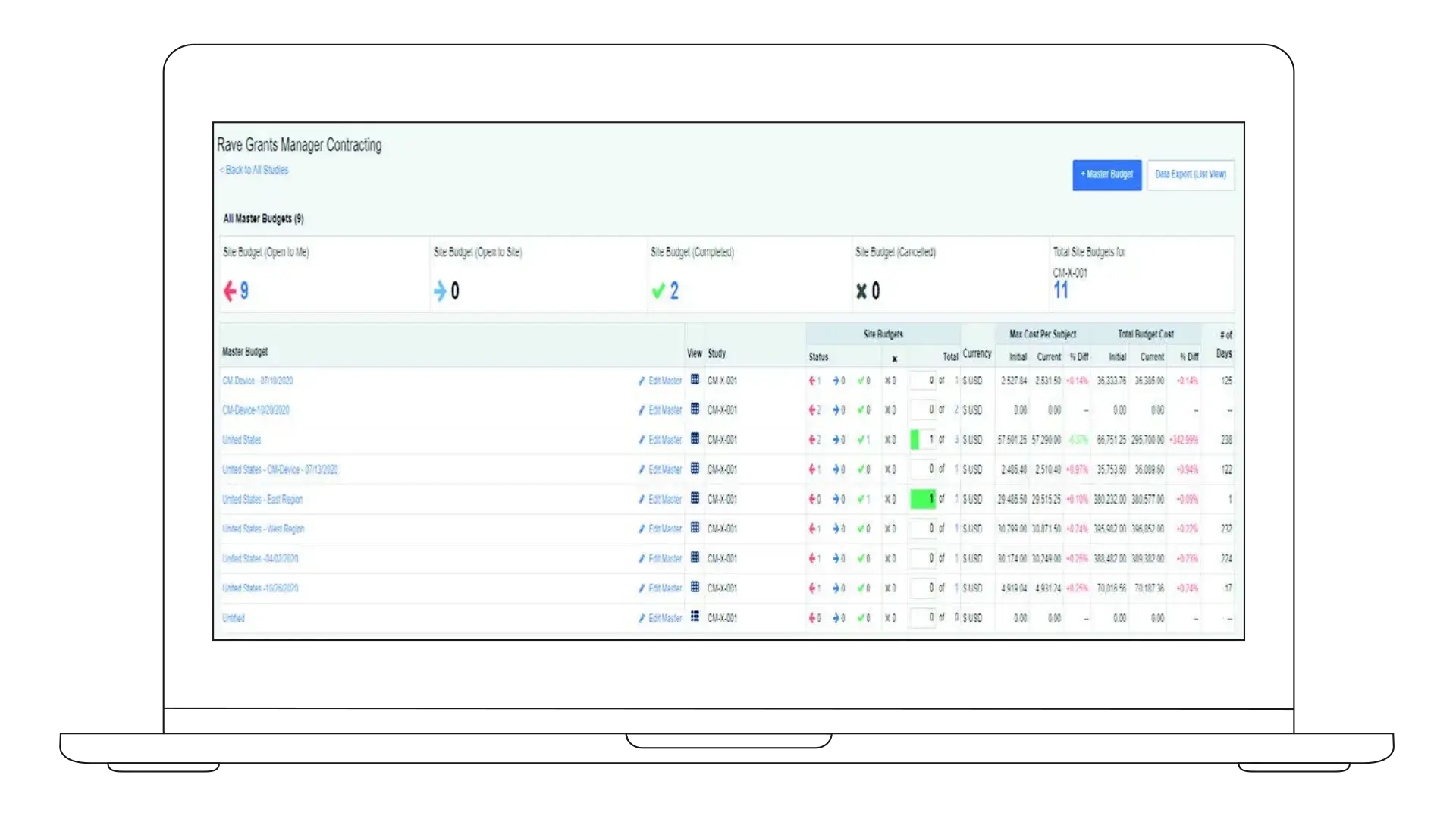
Medidata Grants Manager brings the investigator grant lifecycle into one workflow, so you can build accurate budgets, negotiate with clarity, and maintain financial control as studies scale.

Build Budgets with Confidence
Budgets only hold up when data and process stay aligned. Grants Manager combines fair market value (FMV) benchmarks and structured workflows to support consistent budget planning and negotiation, giving teams a clear rationale behind every decision.
Accurate Budgets from the Start
Fair Site Budgets That Build Trust
Faster, Clearer Negotiations
Financial Oversight as Studies Scale
AI-driven Forecasting
Use Real Trial Data across the Grant Lifecycle
Accelerate Timelines
Build Budgets on Active FMV Benchmarks
Grants Manager supports investigator budget planning using FMV benchmarks informed by real trial grant data.
Benchmarks reflect procedure type, geography, and study context, designed to reflect real conditions.
Teams start from budgets that are grounded in data and built to endure beyond initial planning.
Featured Resource
Better Data, Better Decisions
As trial complexity grows, cost management needs to be precise and defensible. See how fair market value grounded in real trial data supports more reliable budgeting and decision-making.
Download White Paper
FAQ
Explore Experiences
Discover the Medidata Platform