Intelligent Trials
Design and Execute Faster Trials with AI-Powered Analytics
Design smarter studies and drive operational performance with Medidata’s industry-wide, real-time, and historical trial data combined with AI-powered analytics. Confidently optimize your protocol to balance scientific goals with operational feasibility, select high-performing sites that support enrollment and diversity targets, and monitor progress to proactively mitigate risks. Intelligent Trials helps you reduce trial burden, minimize delays, and improve execution from start to finish.
Benefits of Clinical Trial Analytics

Design Operationally Feasible Studies
Design smarter studies by leveraging cross-industry benchmarks and predictive modeling to evaluate protocol decisions before your trial begins. Simulate design changes to understand their impact on enrollment, retention, cost, site burden, and patient burden, so that you can strike the right balance between scientific goals and operational success.
Identify Optimal Study Footprint
Assess historic enrollment rates from industry-wide trials to determine realistic timelines and study footprints. Then, analyze which countries and sites would be optimal for your study’s goals and timelines.

Select High-Performing Sites
Confidently select the right sites for your trial and target patient populations using AI-powered forecasting that uses actual site-level enrollment and congestion data, delivering greater accuracy and reliability for study planning.
Learn more about Intelligent Trials Study Feasibility.

Enroll More Diverse Patients
Find sites with a proven track record of recruiting diverse patients to ensure your trials reflect the diversity of real-world populations and meet regulatory diversity requirements. Our indication-specific, cross-industry patient demographics data allows you to identify and prioritize sites that consistently meet diversity goals, helping you achieve your inclusivity targets and drive equitable clinical outcomes.
Learn more about the Intelligent Trials Study Feasibility Diversity Module.
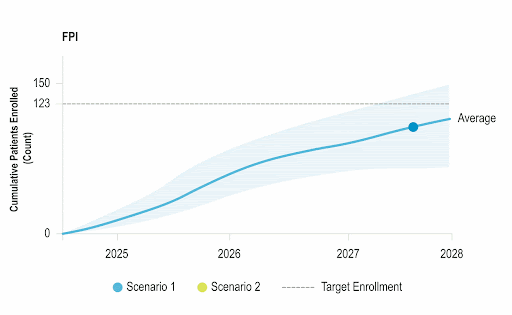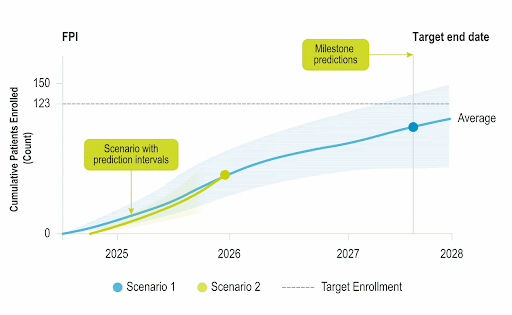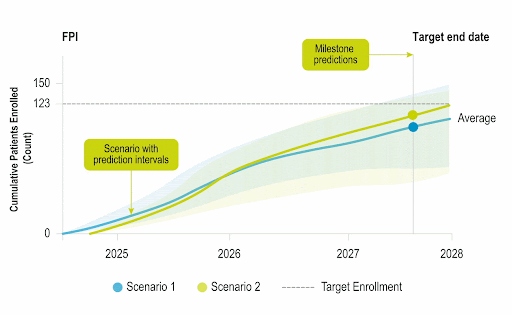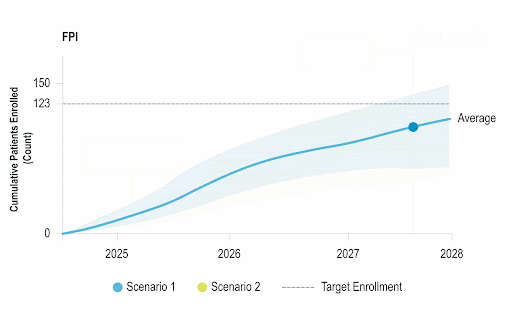
Stay Ahead of Enrollment Risks with Live Study Forecasting
Improve your forecasting accuracy by evaluating the progress of active studies using current performance data and industry benchmarks. Use near-real-time insights to monitor study health and detect enrollment risks early before they threaten your timelines
Key Features

One of the Industry’s Largest Clinical Trial Datasets
Gain insights from cross-industry clinical trial data spanning 36,000+ trials and 11 million patients across over 140+ countries to design and run more effective studies. Our AI-powered solutions transform site-, patient-, and indication-level data into clear, actionable insights, helping you model scenarios, mitigate risks, and make confident decisions at every stage of your trial.
Unmatched Site-Level Granularity
Utilize true site-level operational performance metrics – including enrollments, screen failures, data quality, competitive trials, and patient diversity– giving your team an unmatched, actionable view of each site’s current capabilities. Pinpoint high-performing sites that will help you hit your enrollment timelines, meet your diversity action plan goals, and give you a competitive advantage in a challenging recruitment landscape.

Actionable Real-Time Insights
Leverage data refreshed weekly from over eight thousand active studies to ensure your decisions are based on relevant, timely insights. This allows your teams to calibrate performance and take immediate action in a changing landscape.

Turnkey with White-Glove Support
Make confident decisions via access to off-the-shelf SaaS solutions, and available APIs combined with dedicated white-glove services support.
Learn More

Featured Whitepapers and Reports
The State of Black Participation in Clinical Trials
In our recent research report, we used Medidata’s industry-leading clinical trial data repository to assess the current state of racial diversity beyond the FDA Snapshot.

Featured Case Studies
Medidata Selected by Worldwide Clinical Trials to Accelerate Trials and Transform Patient Experience
Medidata and Worldwide Clinical Trials have expanded their partnership to leverage Medidata’s advanced solutions and extensive data set, enhancing Worldwide’s clinical operations with data-driven insights to accelerate trial timelines, improve site selection, and advance the life sciences industry.

Featured Podcasts and Webinars
ASCO Education: Racial Disparities in Clinical Trial Participation
Learn about the state of clinical trial diversity in clinical trials and what clinicians and trial sponsors can do to improve participant diversity.

Featured Blogs
3 Ways to Approach Challenges During Study Feasibility Processes
Industry data and predictive models, in real time, enable pharmaceutical companies and CROs to improve clinical trial site selection, forecast enrollment timelines, and optimize study feasibility through accurate scenario analysis and site performance predictions.