Coder
Manual medical coding delays database lock and increases the risk of inconsistency across studies.
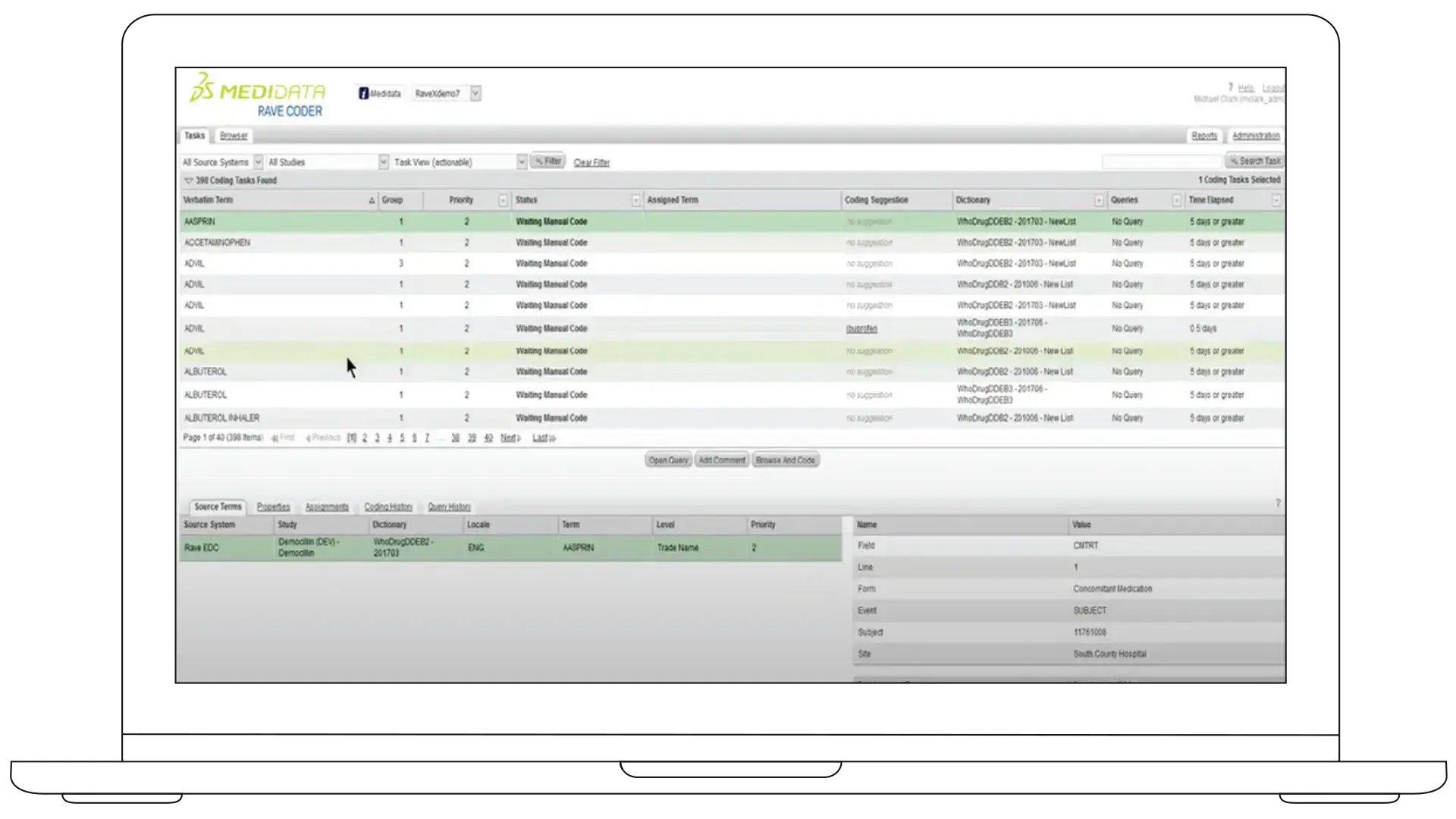
Medidata Rave Coder delivers fast, accurate coding for verbatim terms from Rave EDC and external sources.
Using machine learning and natural language processing, it automatically suggests the most appropriate dictionary matches—so your coders stay in control while working more efficiently.

Work Smarter across Every Coding Task
You need medical coding that's faster, more accurate, and easier to manage across studies. Coder helps your team reduce manual effort, maintain consistency, and stay current without adding operational burden.
Coding Suggestions Algorithm
Dictionary Maintenance and Up-versioning
Synonym List
Consolidated Query Management
External Verbatim Term Management
What Teams Gain with Coder
Accurate Coding
Accurate Coding You Can Trust
You need coding decisions you can stand behind—without slowing your study down. Coder automatically identifies the closest matching dictionary terms using natural language processing and machine learning–driven coding suggestions.
As your study progresses, the algorithm learns from coding decisions to continuously improve match quality—helping you increase consistency, reduce rework, and maintain confidence in your coded data.

Explore Experiences
Discover the Medidata Platform

