Clinical Data Studio
Clinical trial teams need to act on signals across many data sources and roles. When work is fragmented, risks surface late and decisions slow.
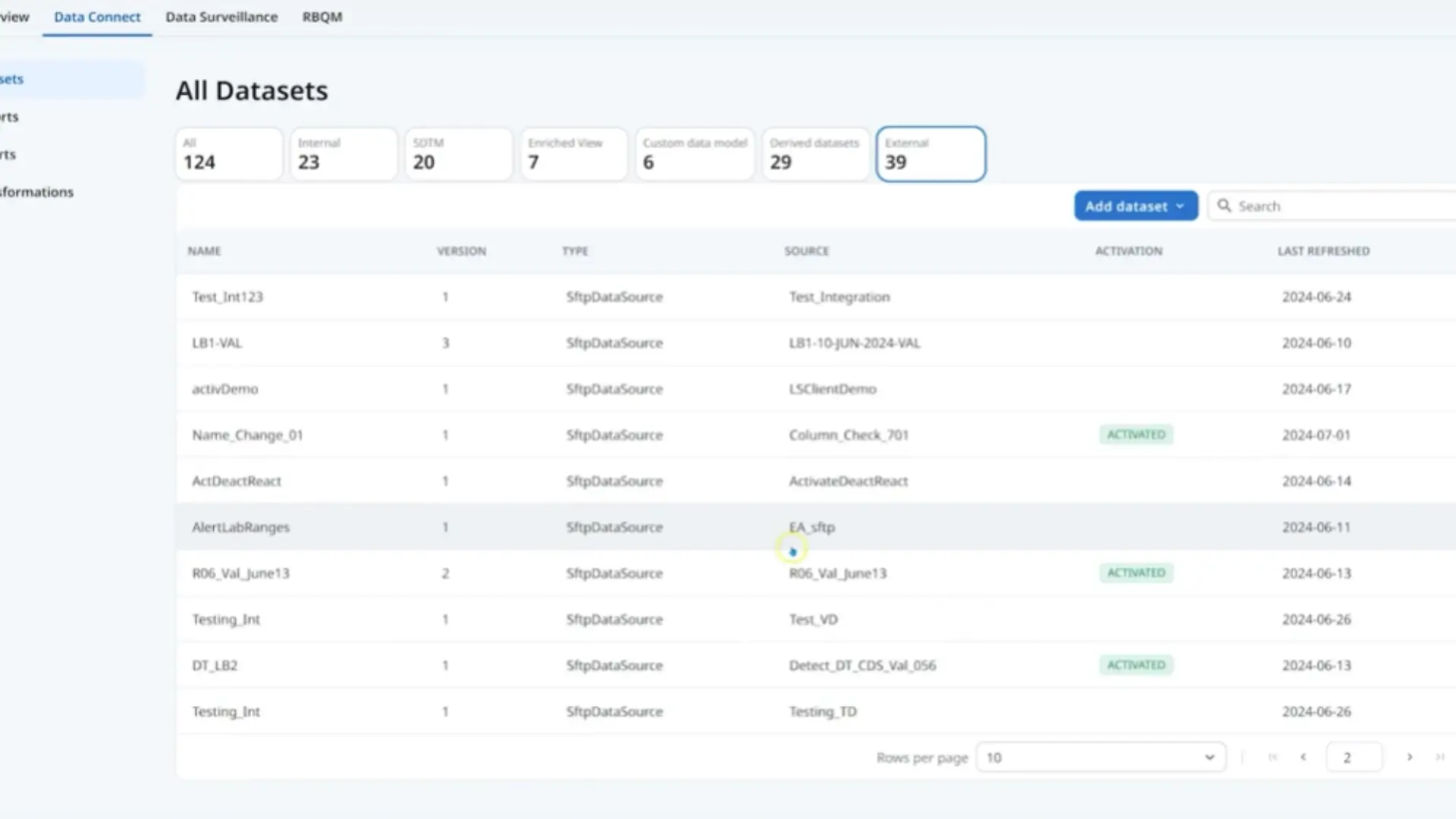
Medidata Clinical Data Studio (CDS) connects data from Medidata and external sources into a single review and monitoring environment to maintain quality and keep trials on track.
Confident Trial Oversight
Core data review supports clinical data management, central monitoring, risk management, and medical monitoring in a single environment. Teams can review data in context, assess risk earlier, and maintain clear oversight.
Instead of moving between disconnected tools, teams work from the same data to support faster, more confident decisions.
Data Integration
Data Review
Patient Profiles
RBQM
Audit Trail Review
The Power of Unified Data
Review Data Faster
Accelerate Data Review and Ensure Data Quality
Review all clinical trial data in one place with AI and automation.
Data managers get access to integrated data sooner and shorten data review cycle times by up to 80% per cycle to deliver cleaner data, faster.

Trusted by Sponsors and CROs
To meet growing data demands and customer expectations for speed and efficiency, we selected Clinical Data Studio.
Evan Hughes
Vice President, Clinical Data Science
...Breaks down data silos and seamlessly integrates into our current software stack, while maintaining quality and integrity across all data sources...
Shobha Dhadda, PhD
Chief Clinical Science & Operations Officer

We can see real-time data and identify any anomalies, track data trends and issues, and resolve those issues for a cleaner database. Clinical Data Studio helps us achieve faster database lock timelines.
Swathi Vasireddy
Associate Director, Clinical Data Management

Featured Resource
Demo: AI-powered Data Quality Management in Action
Explore how Clinical Data Studio integrates data, supports real-time review, and reduces technical burden through its low-code, AI-enabled environment.
Watch VideoFAQ
Explore Experiences
Discover the Medidata Platform