Decentralized Clinical Trials
Decentralized trials promise flexibility and speed—but delivering it without compromising safety, data quality, or site workflows is a real challenge.
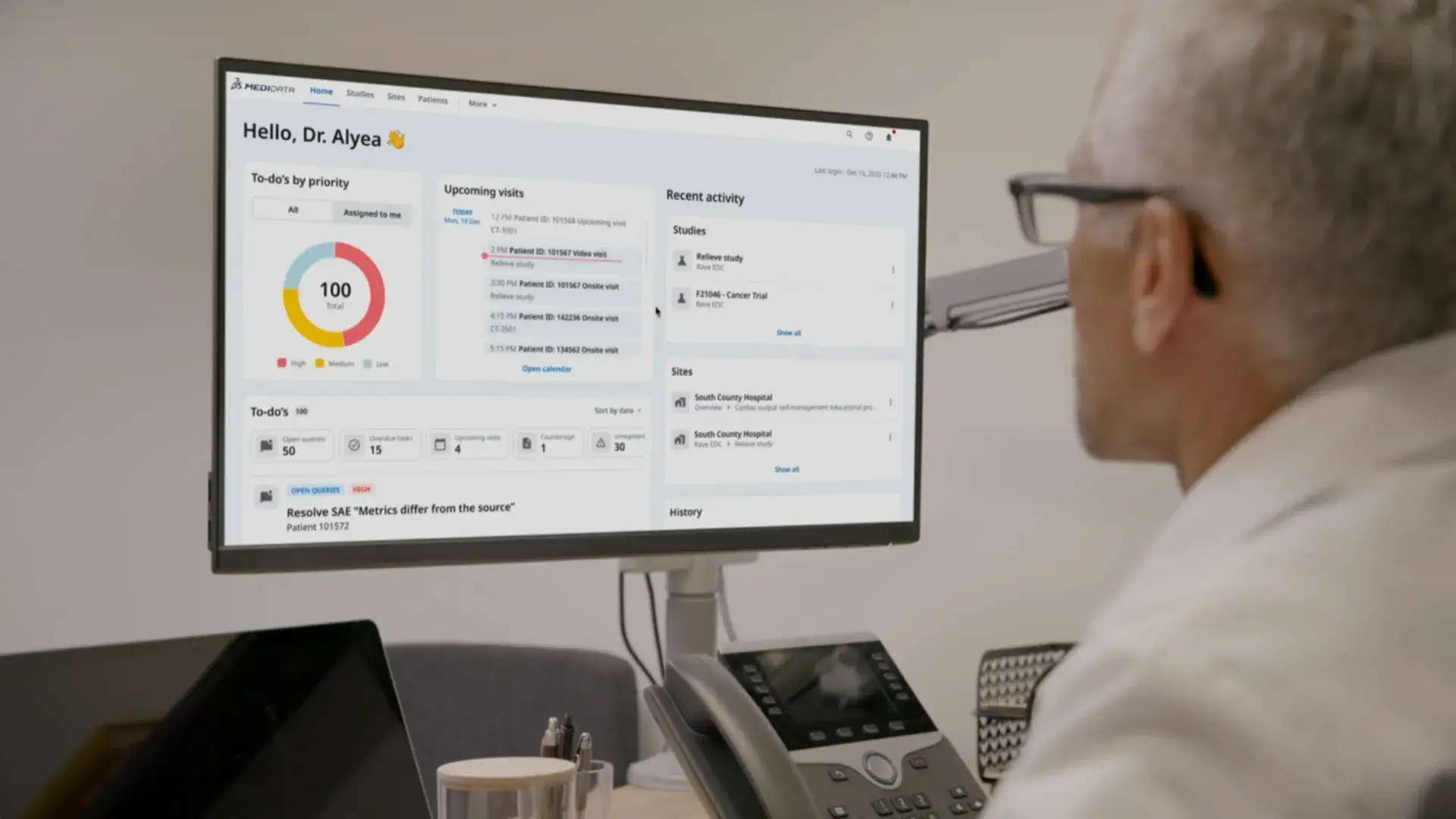
Medidata’s integrated Decentralized Clinical Trials (DCT) program helps teams run hybrid and fully remote trials that work for patients, sites, and sponsors alike—using Medidata Consent, eCOA, and Sensors solutions designed for real-world execution.
Run Your DCT
from
One Platform
Decentralized trials place new demands on patients and sites. From unfamiliar technology to coordinating drug delivery and lab collection, teams must still maintain compliance and data quality.
You need a solution that supports it all, simply and in one place.
Suite of Patient Engagement Solutions
Continuous Clinical Data Monitoring
Direct-to-patient (DtP) Investigational Product Delivery
Circuit Clinical Partnership for Turnkey Solution
Flexible, Custom Design with Medidata’s Trial Dial™
Built to Ease Complexity for Everyone
Sponsors
For Sponsors:
Control and Confidence
Centralized oversight helps sponsors maintain compliance, data integrity, and patient safety—while scaling decentralized, hybrid, or fully remote trials with confidence.
Download Fact Sheet
FAQ
Explore Experiences
Discover the Medidata Platform